
The less directional, more diffuse d and f orbitals, in which unpaired electrons reside, overlap less effectively, form weaker bonds and thus dimerisation is generally disfavoured. In contrast, radicals in d- and f-block chemistry are very common. Radicals can be stable if dimerisation would result in a weak bond or the unpaired electrons are stabilised by delocalisation. These orbitals are strongly directional and therefore overlap to form strong covalent bonds, favouring dimerisation of radicals. Radicals are uncommon in s- and p-block chemistry, since the unpaired electron occupies a valence p orbital or an sp, sp 2 or sp 3 hybrid orbital. In organic chemistry they typically only occur briefly during a reaction on an entity called a radical however, they play an important role in explaining reaction pathways. As the formation of electron pairs is often energetically favourable, either in the form of a chemical bond or as a lone pair, unpaired electrons are relatively uncommon in chemistry, because an entity that carries an unpaired electron is usually rather reactive. 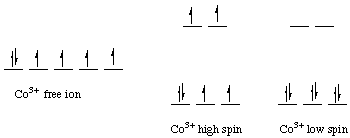
Each atomic orbital of an atom (specified by the three quantum numbers n, l and m) has a capacity to contain two electrons ( electron pair) with opposite spins. In chemistry, an unpaired electron is an electron that occupies an orbital of an atom singly, rather than as part of an electron pair. 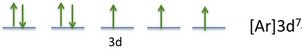
Type of lepton that orbits an atom on its own Periodic table with elements that have unpaired electrons coloured
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
